Turning a specialist task into actionable engineering insights
One moment that has really stayed with me was when I was watching Chief Developer in ThermoPhys Dr. Ailo Aasen explain passionately about energy efficiency to King Willem-Alexander and Queen Máxima of the Netherlands.
His enthusiasm was unmistakable and contagious. It certainly stuck with me, and perhaps with the royal family as well. And rightly so. To stay on track with global climate targets, energy efficiency must improve by around 4% per year according to IEA, more than twice the current pace.
The obvious questions are: How do we achieve this? And why haven’t we done more already?
In this article, I’ll try to shed some light on this.

What is exergy and why does it matter?
Waste heat and electricity from the socket both carry energy. Waste heat can be used to heat homes in the winter. Electricity can also provide heat, via a heater or, better, a heat pump, but it can also be used to charge your phone or power a motor.
Intuitively, we know that waste heat cannot efficiently charge a phone. But let’s imagine an imaginary process that converts the waste heat into electricity while fully respecting the laws of thermodynamics. How much electricity could we actually produce?
That is exactly the question exergy analysis answers.
Exergy measures the ability of different forms of energy to do useful work. It recognizes that electricity and low-temperature heat may contain the same amount of energy, but the energy has different abilities to perform useful work. Any serious discussion about energy efficiency in industrial processes therefore needs to be grounded in exergy, not just energy balances.
So why isn’t this the case?
Exergy analysis has been a specialist activity – until now
The short answer: because it’s painful.
Exergy analysis has not been natively supported and integrated in commercial process simulators. Engineers typically need to export stream data, extract thermodynamic properties, and then post-process everything manually in Excel or Python to calculate the exergy destruction for each unit operation.
And even that’s not the whole story. An important component of the total exergy called chemical exergy, is looked up in tables in textbooks. Yes, textbooks. In 2026.
Those table values are usually defined at 25 °C. In Norway, 25 °C is a rare summer luxury. A much more realistic ambient temperature for most of the year is around 10 °C, and ambient conditions matter a lot in exergy analysis. Changing them affects all results, and therefore also the conclusions.
With today’s situation, changing the ambient temperature in exergy analysis requires going back to a handful of exergy specialists, the ones with the biggest glasses (like Ailo).
Once performed, the results are easy to interpret
Ironically, while exergy analysis is hard to perform, the results are usually straightforward to interpret.
You need an expert to do the analysis, but once it’s done, most engineers can quickly see where the real inefficiencies are and make good assessments to how the process can be improved.
Let’s look at a relevant example.
How TP-Process changes the game and makes exergy analysis easy
The equations behind exergy analysis are well defined. In principle, the entire workflow can be automated.
That is exactly what we have done in TP-Process.
Exergy analysis is now native, automatic, and available for any process simulation. No exports, no spreadsheets, no PhD required just to get the numbers.
To illustrate this, let’s look at a simplified liquid CO2 conditioning process. This is an energy-intensive system with significant potential for improvement, making it a good example.
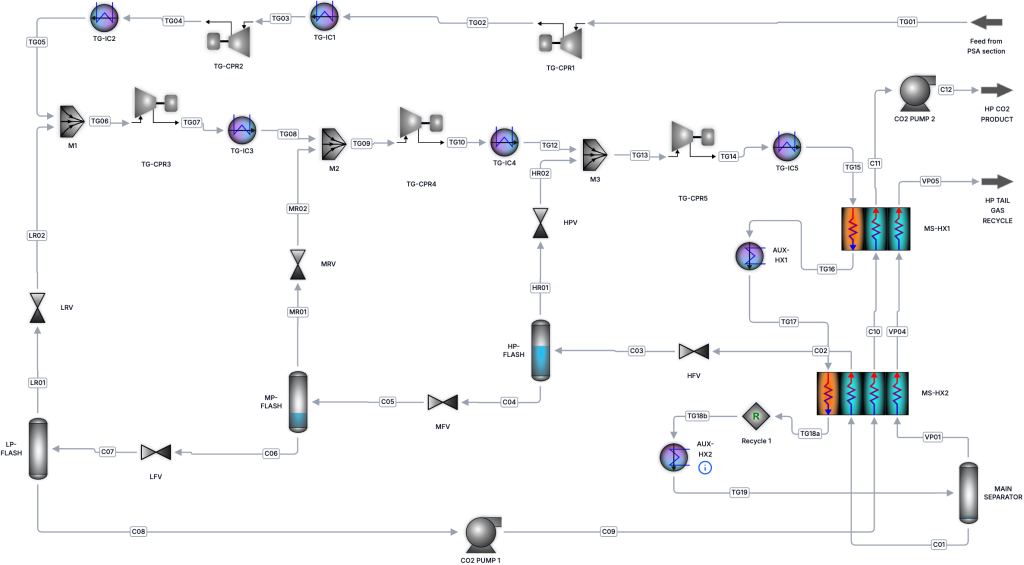
When we solve this process and then open the results view and select Exergy, we immediately get the full breakdown of exergy destruction by unit.
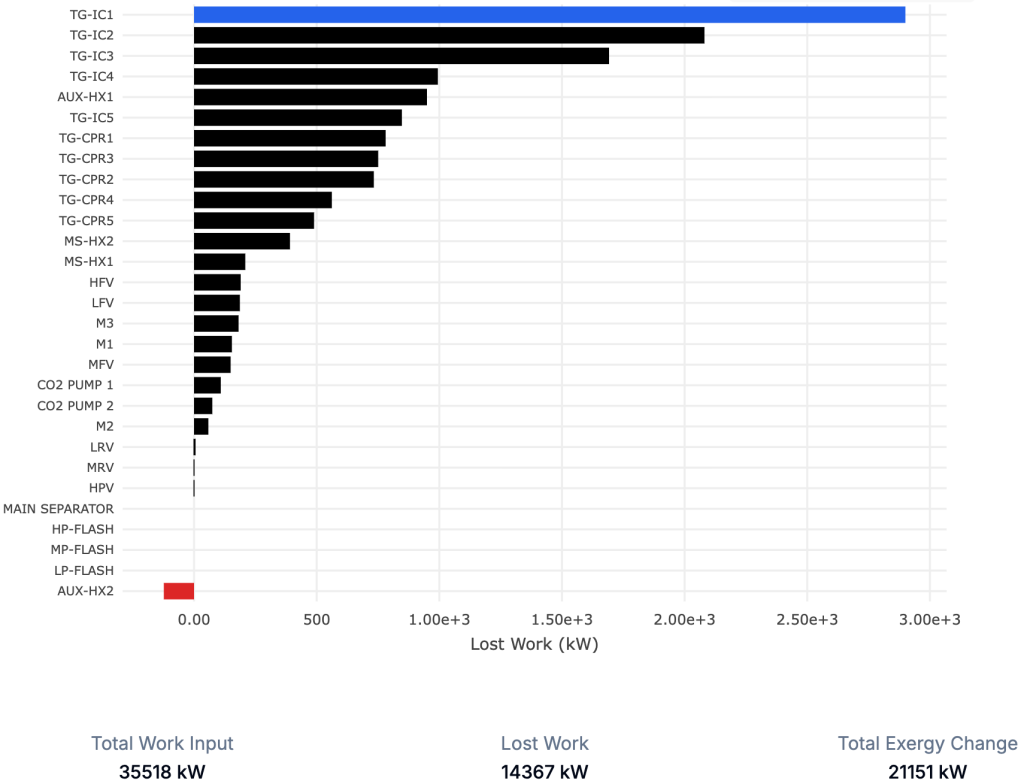
The “magic” here is that all of the exergy destruction, together with the exergy added to the inlet stream, adds up exactly to the total work demand of the process. This means that if we want to reduce the work demand and thus the operating cost, we need to reduce the size of the bars representing lost work.
In this process, we notice something interesting: the heater “AUX-HX2” has a negative exergy destruction, which is physically impossible. This is a clear signal that some of our assumptions are unrealistic.
In this case it turns out that we specified a non-physical coefficient of performance (COP). After specifying a realistic efficiency for the cooler, we obtain a consistent result, and we also realize that the overall work demand increases. That’s valuable insight in itself.
Now it becomes clear that the coolers (“TG-IC1”, “TG-IC2”) are responsible for most of the lost work. As engineers, we immediately start thinking about mitigation strategies – option A, B, or C – rather than spending time calculating exergy numbers.
And if you’re working in Norway (or anywhere else), updating the ambient conditions to get a location-specific analysis is just a few clicks away.
Key takeaways
- Exergy analysis is native, automatic, and integrated in TP-Process
- The results are easy to interpret once they are available
- Poor assumptions are immediately revealed (e.g. negative exergy destruction)
- Ambient conditions can be changed easily and consistently
This functionality will help making exergy analysis a standard and frequently used part of process design, and we hope that it will contribute to improving energy efficiency at the scale required to meet climate targets.